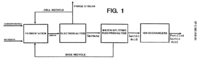
Polylactic acid/Research Development
We are like dwarfs on the shoulders of giants, so that we can see more than they, and things at a greater distance, not by virtue of any sharpness of sight on our part, or any physical distinction, but because we are carried high and raised up by their giant size.
Literature review
Below is a review of peer reviewed publications and expired patents covering the areas of lactic acid from microorganisms, purification of lactic acid to polymerization grade quality, and methods of a polymerization of polylactic acid. An effort was made to reference sources that are freely available online outside of subscription databases, but in certain areas of recent progress sources requiring subscription are used. The information below informs the rationale of the proposed manufacturing protocol.
Process Reviews
L (+) lactic acid fermentation and its product polymerization
L (+) lactic acid fermentation and its product polymerization (2004) by Narayanan et al reviews the production of lactic acid and its use as a plastic monomer. The synthetic route of lactic acid is four steps that involve fixating an activator cyanide group to an acetylaldehyde to form lactonitrile, hydrolysis of lactonitrile with sulfuric acid to yield lactic acid and ammonium salt. For purification via reactive distillation lactic acid is esterified with methanol to methyl lactate and water, methyl lactate is distilled, and hydrolyzed to lactic acid with the addition of water. The production of lactic acid from biological sources is through the fermentation of high energy carbohydrates to lactic acid by Lactic Acid Bacteria. Lactic acid is neutralized and precipitated with calcium hydroxide. Calcium lactate is collected and hydrolyzed with water. For purification lactic acid is esterified with methanol to methyl lactate and removed via distillation, before hydrolysis with water. Measurement of lactic acid can be obtained by HPLC, NAD+ colorimetric assay, or gas chromatography, in order of preferability.
Lactic acid bacteria have been extensively studied, particularly lactobacillus used in dairy preparation and lactobacillus species used for lactic acid production include Lactobacillus delbreuckii subspecies bulgaricus, Lactobacillus helveticus, Lactobacillus amylophylus, Lactobacillus amylovirus, Lactobacillus lactis, Lactobacillus pentosus. Rhizopus oryzae are also stereoselective LAB as well as yeasts such as Saccharomyces cerevisiae and Kluyveromyces lactis and have been investigated for their usefulness. Lactase enzymes are stereospecfic and heterolactic species have two isoforms, some species induce their second enzyme only under high concentrations of lactic acid. Certain species also contain allosterically regulated as well as unregulated isoforms. Genetic engineering on lactobacilli has shown success in controlling stereospecficity of products, reaction rate and yield; Rhizopus oryzae mutants are also under study. Favorable feedstocks are high sugar or starch plants. Nitrogen sources represent a major cost to the industry with yeast extract demonstrating superior performance to cheaper alternatives. Techniques to increase yield include pretreatments, simultaneous saccharification, and nutrient supplementation (especially nitrogen - yeast extract).
Different bioreactor configurations have been studied and batch-wise and continuous reactor sketches are provide. Lactic acid fermentation is inhibited by increasing lactic acid concentrations and methods of filtration or pH control by alkali addition are utilized to increase yield. Methods to remove lactic acid product from the fermentation batch include ultrafiltration, ion-exchange resins, and electrodialysis. Continuous cell recycle reactors have shown high performance and utilize membranes to retain cells while removing media. Cell immobilization by biofilm establishment shows higher performance to free floating culture systems. High cell concentrations make it much more difficult to maintain optimal conditions in all parts of the reactor and can stress the cells causing stereoisomerization. Various configurations using plastic chips to increase surface area but gas exchange is a major issue. R. oryzae have a mycelium form which further complicates agitation and gas exchange.
Purification is the major challenge to lactic acid fermentation production and a variety of schemes including membrane separation, solvent extraction, and vacuum distillation. A solvent extraction using a volatile amine weak base (VAWB) is suggested by the author but consumes the volatile reagent during the purification. Table 2 contains information on the various reported polycondensation procedures. Successful polycondensation depends on the proper selection of an azeotropic solvent. Superior polymer properties are reported for ring opening polymerization including higher molecular weight by an order of magnitude, monomer conversion, linearity of product. A catalyst of f 0.05% stannous octoate is suggested. Various processes to increase the sustainability of the process has been investigated including alternatives to salt addition, intensification of membrane configuration, and selection of benign solvents.
Polylactic acid technolgy
Polylactic acid technolgy by Henton (2005) reviews production, purification, and polymerization. Discusses Cargill Dow's plant which is the largest producer at 400,000,000 lb PLA per year and produces over half the market. The plant uses continuous fermentation, preliminary lactide production followed by purification with vacuum distillation and catalytic ring opening polymerization with a tin catalyst. Purification technologies utilize a variety of characteristics of lactic acid to separate it from the broth including filtration, electrodialysis, ion exchange, distillation, liquid and solid extraction, and esterification. Tin octoate is the basis catalyst for lactide polymerization which converts LA to stereospecific form. PLA characteristics include crystallinity which affects Tg and Tm. Life cycle analysis of a accounts for the energy, wastes, and emissions produced in a process and upstream and downstream to measure the sustainable efficacy of the process.
Development of Four Unit Processes for Biobased PLA Manufacturing
http://www.isrn.com/journals/ps/2012/938261/ Development of Four Unit Processes for Biobased PLA Manufacturing by Chae Hwan Hong, Si Hwan Kim, Ji-Yeon Seo, and Do Suck Han (2012) obtain lactic acid from E coli and purify and polymerize polylactic acid on a pilot scale. They divide the process into four steps fermentation, separation, lactide conversion, and polymerization. Fermentation is performed by E coli KCTC 2223 on LB media in shaking flasks or a bioreactor, with fermentation pH maintained at 6.4 by addition of ammonium hydroxide. A maximum yield of approximately 50% of the input glucose at 60 g/L of D-lactic acid was obtained. Separation was performed by stacked electrodialyis to obtain concentrated ammonium lactate, followed by water splitting electrodialysis using bipolar and ion specific membranes to obtain pure lactic acid. To polymerize the lactic acid a two step process of lactide production followed by high molecular weight polymerization. Lactide formation started with removal of free water at 85°C in vacuum followed by addition of catalysts (zinc oxide or Sn(OEt)2) with a mass fraction of 1~5%. The temperature was raised to 150°C and maintained until no more water was produced, and the temperature was raised to 235 C for an hour. Zinc oxide was found to catalyze high molecular weight oligomers 7760 g/mol with a yield of 85% in a short period. A ring opening procedure started with drying the lactide at 60 C followed by catalytic polymerization with ~250 ppm tin(II)bis(2-ethylhexanoate) at 180°C and 1 hr was found produce ~150 g/mol polylactic acid.
A Literature Review of Poly(Lactic Acid)
A Literature Review of Poly(Lactic Acid) (2001) by Donald Garlotta.
Alternative routes
Microbial is an established and viable route to lactic acid production that is an alternative to the petroleum based racemic producing process, however there are other routes using further engineered organisms or catalyzed direct reactions.
Engineering a Cyanobacterial Cell Factory for Production of Lactic Acid
Engineering a Cyanobacterial Cell Factory for Production of Lactic Acid (2012) by S. Andreas Angermayr, Michal Paszota, and Klaas J. Hellingwerf transforms Synechocystis PCC6803 with Bacillus subtilis L-lactate dehydrogenase and transhydrogenase. Expression of the transhydrogenase is deleterious, but coexpression with dehydrogenase mediates the effect and increases lactic acid production. NADH is used as the hydrogen donor and the transhydrogenase is selected to increase the concentration relative to NADPH.
Catalytical conversion of carbohydrates in subcritical water: A new chemical process for lactic acid production
[http://144.206.159.178/FT/616/601140/12505447.pdf Catalytical conversion of carbohydrates in subcritical water: A new chemical process for lactic acid production] investigates the effects of different salts on the conversion of hexose and triose under subcritical aqueous conditions with a focus on lactic acid production. Fructose is found to be superior to glucose with a conversion of 48%, while trioses had higher conversion rates, the highest being dihydroxyacetone with 86% (g g−1) conversion. Catalysts tested include Co(II), Ni(II), Cu(II)and Zn(II), with Zn(II) being superior and utilized as ZnSO4. Temperature, and residence time were altered in a range from 200 to 360 C and residence times from 3 to 180 s, and pressure was kept constant at 25 MPa. The experimental setup used two stainless steel reactors (tube 1: i.d. 1.0 mm, length 700 mm, volume 0.55 cm3; tube 2: i.d. 3.0 mm, length 700 mm, volume 4.95 cm3), with temperature control from a heat block and with flux being controlled by an upstream HPLC pumping in substrate. Downstream of the reactor is a heat exchanger to dissipate heat, spill valve to relieve pressure, and three way valve to direct the process stream to waste or product containers. Conversion, yield and selectivity, are calculated. Fructose can be completely degraded within 2 min at 260 C over ZnSO4 catalyst and the time further decreased with an increase in temperature, with 300 C completing conversion within 20 s. Furthermore selectivity increases from ~35% to 48% when temperature is increased from 260 C to 300 C.
Feedstocks
Lactic acid production by microorganisms is based on conversion of sugars to a lower energy product (lactic acid) due to an unavailability of oxygen for respiration. Feedstocks containing sugars can come from a variety of sources, but ideally it should not compete with food crops. Sources of sugar rich material that do not compete with food crops include sorghum, degraded lignocellulose, and alternative sources such as lipid extracted algal cake. Lignocellulose contains a large amount of fixed carbon and is available in large amounts as the vegetative tissue of harvested crops but is recalcitrant to degradation. A wide variety of feedstocks can be used and must be optimized to an individual situation.
Carbohydrates crops
Optimization of Lactic Acid Production from Cheap Raw Material: Sugarcane Molasses
Optimization of Lactic Acid Production from Cheap Raw Material: Sugarcane Molasses (2012) by Umar Farooq, Faqir Muhammad Anjum, Tahir Zahoor, Sajjad-ur-rahman, Muhammd Atif Randhawa, Anwaar Ahmed, and Kashif Akram compared lactic acid production of lactobacillus delbrueckii using sugar cane molasses as a feedstock over a range of temperatures and feedstock concentrations. The important characteristics of a feedstock for lactic acid are low cost, minimum contaminants, rapid fermentation rate, high lactic acid production yields, little or no by-product formation and year round supply. The agricultural byproduct of sugar cane includes a molasses that contains 45-60% sugars including sucrose, glucose, and fructose. Lactobacillus delbrueckii was isolated from an indigineous yoghurt population by procedure by Harrigan (1998). Fermenation was carried out at 34ºC, 38ºC and 42ºC with 0, 6, 12, 18 and 24% substrate levels without apparent pH control for 7 days. The media contained (g 100mL-1); peptone 10.0, meat extract 10.0, yeast extract 05.0, Tween-80 01.0, K2HPO4 02.0, Sodium acetate 05.0, tri-ammonium citrate 02.0, MgSO4.7H2O 0.2, MnSO4.4H2O 0.05. Total sugars and lactic acid were measured over the experiment on a 24 hr basis. The highest temperature of 42 C was the most productive. The second highest substrates concentration, 18%, achieved the highest lactic acid concentration 11.27 g/ 100 ml, yield 85%, and productivity achieving peak sugar usage on the 3rd day.
Lignocellulose
Fermentable sugars by chemical hydrolysis of biomass
Fermentable sugars by chemical hydrolysis of biomass (2010) by Joseph B. Binder and Ronald T. Raines utilized an ionic liquid, 1-ethyl-3-methylimidazolium chloride (Emim)Cl, with an acid catalyst, HCl and H2SO4 to demonstrate efficient hexose and pentose release from cellulose and lignocellulose biomass. The process is further improved through the slow addition of water to drive the reaction toward glucose formation over degradation products despite the hydrophobicity of cellulose. The corn stover was first treated with Emim before addition of HCl and water. With the addition of water being gradually increased to 43% over 1 hour the glucose yields were increased to 90%. The hydrolysate was run over an ion-exchange column and the resulting conversion product was used as a feedstock for E coli and yeast and performed equitably with an insignificant trend towards better performance by the hydrolysate under low oxyygen conditions. The major cost of the process is in the column chromatography step. The authors speculate on better performing biocatalysts that utilize pentoses (such as Bacillus coagulans).
A Study of the Acid-Catalyzed Hydrolysis of Cellulose Dissolved in Ionic Liquids and the Factors Influencing the Dehydration of Glucose and the Formation of Humins
[http://www.cchem.berkeley.edu/atbgrp/files/ChemSusChem%202011%204%201166.pdf A Study of the Acid-Catalyzed Hydrolysis of Cellulose Dissolved in Ionic Liquids and the Factors Influencing the Dehydration of Glucose and the Formation of Humins]
Hydrolysis of lignocellulosic materials for ethanol production: a review
[http://stl.bee.oregonstate.edu/courses/ethanol/restricted/SunCheng2002.pdf Hydrolysis of lignocellulosic materials for ethanol production: a review] (2002) by Ye Sun, Jiayang Cheng.
Detoxification of dilute acid hydrolysates of lignocellulose with lime
Detoxification of dilute acid hydrolysates of lignocellulose with lime
Microalgae
Nannochloropsis salina biomass to lactic acid and lipid
Nannochloropsis salina biomass to lactic acid and lipid (paywalled) by Talukder and Wu examines the suitability of oleaginous Nannochloropsis algal cake after lipid extraction for lactic acid production by Lactobacillus pentosus.
Lactic acid bacteria
Choice of lactic acid producing microorganism must take into account a number of factors including productivity, stereoisomer production, feedstock flexibility, difficulty culturing - including contamination. Lactic acid production for fermentation is not an uncommon capability including human muscle tissue. A number of microorganisms with prodigious lactic acid production have been isolated and characterized and include well-known members of the lactobacillus genus such as acidophilus and delbrueckii, bacillus bacteria such as bacillus coagulans, and some fungi such as Rhizopus oryzae. Choosing an organism and strain that with high lactic acid productivity needs to consider the molecular information available on these strains as many are under intense study. Lactobacillus exist as many well established strains that are well characterized from their use in yogurt making and early adoption to the lactic acid production industry, while bacillus coagulans has recently been identified in a number of environmental isolation efforts and is now under intense study with multiple strain genomes being recently sequenced. Organisms are available with a variety of licenses from biological specimen supplier American Type Culture Collection (ATCC).
Bacillus coagulans
Bacillus coagulans has been an organism of recent research focus due to its possible applications in biomass conversion. To further this work genome sequencing has been performed by American and Chinese research groups e.g. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3905273/ and draft sequences have been published http://www.ncbi.nlm.nih.gov/genome/?term=CP002472 JGI. There is a D-LA dehydrogenase gene present that functionally expresses in E coli, but there is no detectable endogenous activity most likely due to nonexpression. B. coagulans has been isolated a number of times from the environment based on lactic acid productivity and it is under intense study for application due to its thermotolerant growth and robustness. Bacillus coagulans thermophilic nature not only removes the necessity of a sterilization of feedstock, but it overlaps with fungal lignocellulases optimal temperature and pH giving possibility to using low value lignocellulose agricultural byproducts. Bacillus coagulans is related to a well studied organism Bacillus subtilis and its molecular biology is being characterized. Plasmid transformation using electroporation has been described and vector sequences for a B coagulans/ E coli plasmid.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3165500/pdf/zjb4563.pdf http://genome.jgi-psf.org/bacco/bacco.home.html
Non-Sterilized Fermentative Production of Polymer-Grade L-Lactic Acid by a Newly Isolated Thermophilic Strain Bacillus sp. 2–6
Non-Sterilized Fermentative Production of Polymer-Grade L-Lactic Acid by a Newly Isolated Thermophilic Strain Bacillus sp. 2–6 by Qin et al examines Bacillus coagulans lactic acid fermentation capacity in batch and fed-batch mode with unsterilized feedstock. The strain was newly isolated and high productivity and optical purity was achieved in both cases suggesting it may be a promising organism for lactic acid fermentation. The productivity obtained are higher than other reported strains. The report obtained 730 isolates at 55 C growth from 7 soil samples and the 2-6 strain was selected as the most productive lactic acid producer. Bacillus coagulans identity was assigned based on 16S rRNA gene sequence and an enantiomeric ratio of very high L:D enantiomers. NAD-dependent lactate dehydrogenase activity could was only detectable for the L-lactate dehydrogenase through active stained native PAGE, demonstrating enzymatic activity is entirely L-lactic specific. Initial screens utilized 97 g/L glucose and higher glucose concentrations were examined for higher productivity. Concentrations of glucose above 133 g/L were found to inhibit glucose consumption and lactic acid productivity and 97 g/L and 133 g/L glucose concentrations were used for further optimization. The nutrient requirements of the strain for nitrogen source and vitamins were investigated and the most cost effective media components of glucose 97–133, YE 12.6, soy peptide 1.2, cottonseed protein 3, NaNO3 1, NH4Cl 1, were found to produce 95% of maximum yield. Two phases of lactic acid production were identifiable in batch mode from 0-15 hour during which cell growth and lactic acid productivity were coupled and 15-30 hours when cell density reached stationary phase and lactic acid production continued to complete consumption of the glucose, albeit at a slower rate. Three fed-batch regimes were tested and the effectiveness of continuous feeding and pulse feeding were found to be superior to exponential feeding. Fed-batch experiments were conducted in 5 l and 30 l volumes with no noticeable differences in rates or products. Analysis of organic acid products using organic acid HPLC columns revealed no other detectable products. Overall, Bacillus coagulans strain sp 2-6 is a highly promising strain for the production of enantiomeric pure lactic acid in an industrial process.
L(+)-Lactic acid production from non-food carbohydrates by thermotolerant Bacillus coagulans
| L(+)-Lactic acid production from non-food carbohydrates by thermotolerant Bacillus coagulans by Ou et al examines the ability of Bacillus coagulans 36D1 to utilize carbohydrates from lignocellulosic materials treated with fungal lignocellulases. Lignocellulose materials could be a low cost source of sugars, but there are several limitations to implementation of an efficient industrial process, mainly the cost of treatment to breakdown recalcitrant lignocellulose material, complex product mixture produced by many microorganisms in industrial use, and low yield of desired products by homolactic organisms. Bacillus coagulans displays traits that may allow it to overcome these hurdles to adoption of biocatalytic lignocellulose lactic acid production. As a thermophilic organism with a growth temperature of 50-55 C and favors slightly acidic conditions lignocellases are not inhibited during simultaneous fermentation allowing a decrease in the amount of costly enzymes. Coagulans consumes the released hexoses and pentoses relieving product inhibition and creating a productive pathway. Coagulans possesses the pentose-phosphate pathway for efficient use of pentose sugars unlike other industrially known strains which possess the phosphoketolase pathway resulting in equimolar production of acetic acid and lactic acid (lowers efficiency and complicates purification). Despite its apparent advantages coagulans titer of lactic acid (depending on the strain and conditions) is often lower than is reported for other lactic acid bacteria. This study utilized salting out of the lactic acid product using calcium carbonate (CaCO3) and demonstrated titers of over 100 g/L.
Engineering Thermotolerant Biocatalysts for Biomass Conversion to Products
Engineering Thermotolerant Biocatalysts for Biomass Conversion to Products a technical report by K. T. Shanmugam, L. O. Ingram & J. A. Maupin-Furlow describes progress on characterizing B coagulans metabolism.
Bacillus genetic transformation
Development of plasmid vector and electroporation condition for gene transfer in sporogenic lactic acid bacterium, Bacillus coagulans
Development of plasmid vector and electroporation condition for gene transfer in sporogenic lactic acid bacterium, Bacillus coagulans (2007) by Mun Su Rhee, Jin-woo Kim, Yilei Qian, L.O. Ingram, K.T. Shanmugam contructs a plasmid Being of the same genus as the laboratory model organism Bacillus subtilis may allow the utilization of a number of laboratory plasmids to be platforms for coagulan optimization. Comparable G-C content appears to be the major limiting factor in interspecies transformation in the bacillus genus.
Interspecific Transformation in Bacillus
Interspecific Transformation in Bacillus by Julius Marmur, Edna Seaman, James Levine
Plasmid maintenance often requires continuous application of a selection pressure or else the unnecessary plasmid is jettisoned or lost during replication. Further research is needed into methods of genomic integration through homologous recombination/ strand break repair integration or protoplast transfer. Alternatively, genetic mutants with interruptions to necessary nutrient pathways can be obtained and the reintroduction of nutrient genes reintroduced as a marker. http://www.google.com/patents?hl=en&lr=&vid=USPAT5843720&id=2eYAAAAAEBAJ
Lactobacillus
Lactobacillus is the lactic acid fermenator used in the production of fermented foods (from yogurt to sourdough) and has been used in the industrial fermentation of lactic acid. It produces a racemic mixture of D/L-lactic acid, but its growth characteristics are well known and its productivity is high enough to be profitable.
Biotechnological Production of Lactic Acid and Its Recent Applications
[http://www.aseanbiotechnology.info/abstract/21021670.pdf Biotechnological Production of Lactic Acid and Its Recent Applications]
Optimisation of media and cultivation conditions for L(+)(S)-lactic acid production by Lactobacillus casei NRRL B-441
Optimisation of media and cultivation conditions for L(+)(S)-lactic acid production by Lactobacillus casei NRRL B-441 (2001) by M. Hujanen, S. Linko, Y. Y. Linko, M. Leisola uses fed batch shaking flask fermentation of glucose with Lactobacillus casei using malt extract as a nitrogen source. Lactobacillus casei was chosen for its favorable sterioisomer ratio and the study aimed to optimize the fermentation process using industrially relevant growth conditions with a low cost nitrogen source. Maximum concentration obtained was 118 g/L from 160 g/L glucose and maximum productivity of 4.4 g/L/h was achieved at 100 g/L glucose at 15 hours fermentation. Malt sprout extract to glucose in a ratio of 53.8:100 was used throughout the study for a standard amount of nitrogen:carbon 22:100. Growth was conducted 1.5 l bioreactors at 35 °C, pH 6.3, 200 rpm, with pH adjustment by NaOH addition. Lactic acid and glucose were determined by HPLC using an Aminex HPX-87H+ cation-exchange column and consumption of glucose and the production of lactic acid was monitored with a YSI 2700 Select Biochemistry Analyzer. Malt sprout extract used as sole nitrogen source was found to show a loss in productivity but could be effectiviely used to supplement reduced concentration of yeast extract, down to 4 g/L. Resting fermentation was also conducted but showed a slight loss in productivity.ided by fermentation time). The maximum productivity with yeast extract was 6.0 g l at 16 h fermentation time compared to 4.9 g/l/g at 16 h by malt sprout extract and resting cells were 3.5 g/l/h.
Comparison of lactobacillus delbrueckii and bacillus growth and lactic acid productivity
Rhizopus oryzae
L(+)-lactic acid production by pellet-form Rhizopus oryzae R1021 in stirred tank fermenter
L(+)-lactic acid production by pellet-form Rhizopus oryzae R1021 in stirred tank fermenter by Bai et experimented with growth parameters effects on culture growth in a continuous run fermentor. Parameters under study included NH4NO3 concentration, CaCO3 addition timing, agitation speed and aeration rate, and inoculation concentration and effects on growth morphology and lactic acid were studied. Pellet form exhibits higher lactic acid productivities and inoculation of 10e6 spores/ml and addition of CaCO3 at 8 hours exhibited pelleted forms. Lactic acid yield was ~72.5% and with 300 rpm and aeration of 0.6 vvm yield increased to 74.5%. Biomass is limited by oxygen transfer, high biomass is necessary for high turnover of glucose to lactic acid. Biomass to lactic acid productivity was found to be highest with 2 g/l NH4NO3, 100 g/l glucose, 300-600 rpm, and 0.6-1.2 vvm. Repeated cycles using the R. oryzae culture showed sustained viability through the 7th cycle and an increase in lactic acid yield to 80%.
Optimization of lactic acid production with immobilized Rhizopus oryzae
Optimization of lactic acid production with immobilized Rhizopus oryzae (2012) by Muhammet Şaban Tanyıldızı*, Şule Bulut, Veyis Selen and Dursun Özer uses polyurethane foam as a matrix and alters basic fermentation conditions.
Lactic acid production and purification
Early purification methods involved precipitation of a lactate salt, filtration, and hydrolysis back to lactic acid. This method had high recovery rates, but it produced large amounts salt waste (calcium sulfate) and consumed large amounts of chemical substrates (calcium hydroxide and sulfuric acid). Alternatively other salts could be used that produce a waste that is easier to handle and recover, and under consideration is the universal base sodium hydroxide.
Lactic acid cannot be easily distilled in high purity, but an ester with a small chain organic alcohol can be separated from impurities over a moderate sized distillation column. Reactive distillation of an ester of methanol and lactic acid over a 20 stage distillation column yields high purity ester that can be hydrolyzed back to the acid form. This process is capital and energy intensive, but it gains efficiency in large scale production. The process should be investigated for feasibility and efficiency on a small scale.
Lactic acid must be purified in high quality from the fermentation media in order to be suitable for polymerization. Advances in purification have involved semipermeable membrane sieves and more recently electrophoresis technology. Electrodialysis has been widely adopted for large scale organic acid production. A commercialized route of lactic acid production for polymerization from fermenation uses conventional electrodialysis to separate and concentrate the lactate salt (a basic waste stream can be recycled to the fermentor), followed by watersplitting ED with bipolar membranes to produce a highly concentrated lactic acid. Another method to eliminate the use of a pH balancing base and the production of a waste salt is size selective microfiltration, ultrafiltration and nanofiltration in a crossflow configuration.
http://www.sciencedirect.com/science/article/pii/S0960852412014460 Open fermentative production of l-lactic acid by Bacillus sp. strain NL01 using lignocellulosic hydrolyzates as low-cost raw material (paywall)
Membrane separation
Process intensification in lactic acid production by three stage membrane integrated hybrid reactor system
Process intensification in lactic acid production by three stage membrane integrated hybrid reactor system describes lactic acid fermention without alkali addition by the constant removal broth and separation through microfiltration and two stage nanofiltration. Innovative use of size /salt selective membranes to separate lactic acid from the fermentation broth utilizes pressure and crossflow filtration to with microfiltration and two stage nanofiltration (NF-1 and NF-2 membranes manufactured by Sepro) to produce high quality (96%) grade lacic acid. The author claims significant gains can be made in productivity and utilized a highly available feedstock of sugar cane water. The physical formulas of crossflow filtration and economic impact of widespread implementation are proposed. Membrane filtration is shown to be less energy demanding in addition to not producing significant waste. Avoiding the phase changes present in the current industrial salting out method leads to membranes significant energetic and capital cost reductions. L(+)-homolactic acid lactobacilli dulbreike is used as a biocatalyst, achieved high yields, and is available from ATCC. The fermentation broth must be sterilized before fermenation and is maintained at 40 C. Procedural techniques also are presented including: a 15 hour lag phase before fermentation broth is started through filtration, and gradual increase in up to ~16 kg/cm2 pressure driving the polishing NF-1 step over the first 12 hours as a means to reject undissociated lactate while allowing lactic acid passage. The nanofiltration stage with NF-2 membranes is conducted at 13 k/cm2. The authors convincingly argue this process intensification can be conducted on a small scale, possibly even based on solar power.
Process intensification in lactic acid production: A review of membrane based processes
Process intensification in lactic acid production: A review of membrane based processes (pay-walled) (2009) by Pal et al reviews membrane based separation advances in lactic acid purification. The broth contents that need to be separated are cells, nutrients, unconverted carbon, water, and lactic acid and membrane based approaches include microfiltration, ultrafiltration, nanofiltration, reverse osmosis and electrodialysis. Separation based on size from larger to smaller pore size is through microfiltration, ultrafiltration, then nanofiltration. Cells and proteins are the main causes of membrane fouling and system design needs to minimize their adsorption. Microfiltration (pores 0.1-1.2 microns) retains cells and allows other broth components to permeate. Nanofiltration (pores no larger than 0.1 nm) has been shown to be able to retain sugars while allowing lactic acid to permeate. Research has focused on coupling these two membranes to effectively separate lactic acid while allowing cell and sugar recycle in a continuous operation fermentor. Pressure is needed to drive flux, low pressure (1–2 kgf/cm2) for microfiltration, and higher pressure (6–15 kgf/cm2) for nanofiltration. PH and cross flow velocity have been identified as a key parameter for fluxes with an increase in both corresponding to an increase in flux. A variety of membrance configurations have been tested including tubular, frame and plate, and hollow fiber modules. The highest performance ultrafiltration unit reported is by Sikder et al with a laboratory-synthesized polysulfone-cellulose acetate blend microfiltration membrane in a cross flow configuration (see below: Synthesis and characterization of cellulose acetate-polysulfone blend microfiltration membrane for separation of microbial cells from lactic acid fermentation broth).
Production and purification of lactic acid
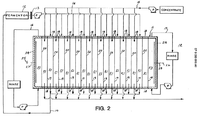
Production and purification of lactic acid European Patent EP0393818A1 issued to Glassner and Datta of the Michigan Biotechnology Institute on October 24 1990 details a commercialized route of lactic acid production from fermentation bacteria. The process uses Lactobacillus acidophilus (ATCC 53681 which cannot be located in the ATCC catalog fed with corn steep liquor and corn oil and reports yields of 80% of the theoretical maximum, with low contamination in the order of less than 1% protein and 10 ppm sulfate ions. The process adds a salt to form lactate complexes which is processed with conventional electrodialysis to create a cell free concentrated lactate salt stream and a dilute broth which is returned to the fermentation chamber. The lactate stream is concentrated through evaporation and processed to a relatively concentrated and purified fraction using water splitting electrodialysis. Strong acid followed by weak base ion-exchange columns are used for polishing to create a purified product of polymerization grade. The process was tested on small 1-2 l, pilot 80 l, and large 500 l scales and productivity and efficiency remained constant as correlated to substrate density and cell density (margin between cell growth versus LA production). The process was tested with continuous batch and cell recycle fermentation. The process is reported to have several advantages including high productivities and recovery rates, low energy requirements (~0.5 kwhr/ lb LA), the possible reuse of salt anion and cation after recovery from the electrodialysis unit, and the dual use of electrodialysis to separate cells from whole broth for recycle without compromising the purity or efficiency of the electrodialysis step. The process details are as follows. A fermentation media containing 1.0-4.0% steep corn liquor and 0.1-1.0% crude corn oil were combined in water, the concentration of carbohydrates is 20-120 g/l and preferably 40-100 g/l. the media is sterilized and inoculated with 5% inoculum. Growth conditions are maintained with agitation at 75 rpm and a temperature between 20-50 C, and preferably 39 C. The media pH is maintained between 4.8 and 5.7 through the addition of carbonates or hydroxides, preferably sodium or ammonium hydroxide. At a cell concentration of 2.5E10-3.0E10 high lactate productivities can be maintained in the order of 2.0-2.5 g/l/hr with a product concentration of approximately 75-90 g/l. Continuous culture fermentation was conducted in seal flasks using a peristaltic pump to remove broth and additions of an equal volume of fresh broth and salts. A cell recycle fermentor consisted of a growth chamber, a 0.2 micron ultra-filtration unit that returned cells to the chamber, and an effluent capturing container. Whole broth was filtered through 200 mesh to remove debris before direct addition to the electrodialysis chamber. The conventional electrodialysis unit consists of 8 stacks of anion and cation permeable membranes with a total surface area of 102.4 cm2. The electrolyte used was 2.5 M NaOH. A representative configuration is displayed in figure 2, however adaptions may be made by the user. The configuration displayed contains distribution flow gaskets (18), anion permeable membranes (19), cation permeable membranes (20), parallel cells (21) consisting of 18+19+20, and end cells (22 and 23) consisting of 19+20+ end caps (24). End cell 22 is connected to the cathode (25) via the a connector (27), and end cell 23 is connected to the anode (26) through a connector (28), both end cells are connected to a rinse system that circulates a electrolyte solution (such as NaSO4 or lactate salt). Fermentation broth is passed through a screen (13) and passed to the electrodialysis unit via line 14, basic solutes are passed through the parallel cells and returned to the fermentor through a continuation of line 14. Line 15 circulates a lactate salt solution in the parallel cells and lactate from the broth is concentrated through the anion permeable membranes (19) and collected on the effluent side of the system. The collected effluent is further concentrated via evaporation.
Process development and optimisation of lactic acid purification using electrodialysis
[http://144.206.159.178/FT/549/63720/1083859.pdf Process development and optimisation of lactic acid purification using electrodialysi] by Madzingaidzo et al examines purification of lactic acid from fermentation broth using mono and bi-polar electrodialysis. A mono-polar membrane selectively allows cations or anions to traverse the layer, while a bi-polar layer is made of a cation and anion membrane that splits water molecules into H+ and OH- for charge balancing. An electrical current is applied to the dialysis chamber to separate molecules according to their charge and mono and bi-layer membranes create channels concentrated with certain components. In mono-layer electrodialysis a alternating semipermeable membranes starting with a cation membrane next to the anode (+), a dilute stream feeds through center from which lactic acid is concentrated through a anion exchange membrane towards the anode. Charge is balanced from an electrode rinse solution that circulates next to the electrodes. A bi-polar uses bi-polar membranes to separate the electrode rinse solution from the concentrating channels creating sections holding a base, salt and final acid form. A measurement of % current efficiency (current used to transport molecule from input to concentrated stream/ total current) is used to evaluate the process.
- Reverse osmosis
Sources still to be summarized
[1] An electrokinetic bioreactor: using direct electric current for enhanced lactic acid fermentation and product recovery
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC203522/pdf/aem00131-0098.pdf Novel Method of Lactic Acid Production by Electrodialysis Fermentation
http://oatao.univ-toulouse.fr/2918/1/Bouchoux_2918.pdf
http://www.sciencedirect.com/science/article/pii/S0011916411010290# Separation of lactic acid from fermentation broth by cross flow nanofiltration: Membrane characterization and transport modelling (paywall)
http://link.springer.com/article/10.1007%2Fs10098-011-0448-z?LI=true (paywall)
Reactive Distillation
Lactic acid purification
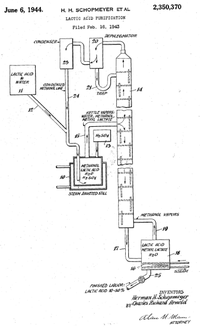
Lactic acid purification issued to Schopmeyer June 6 1944 covers a method to purify lactic acid from fermentation broth by the use of calcium carbonate salts and esterification with methanol for fractional distillation. The fractional distillation set-up includes a boiler containing methanol and lactic acid and a catalyst (H2SO4) that delivers vapors to a fractionation column that allows the separation of a concentrated lactic acid liquor of ~10-50%. The salting out of calcium lactate uses calcium sulfate which is concentrated and converted to acid form through treatment with sulfuric acid, calcium sulfate forms an insoluble fraction. The concentration was usually 40-60% lactic acid and ethanol can also be used as the esterification. The esterification procedure uses a steam jacket with the following substrate mole ratio 1.5:1:0.005 methyl alcohol: lactic acid: sulfuric acid. Start-up of purification uses 80% lactic acid substrate (depending on concentration), 20 methanol, and a small amount of catalyst. Steady-state is maintained by addition of substrates and catalyst, and recycling of methanol.
Optimization of Batch Reactive Distillation Process: Production of Lactic Acid
Optimization of Batch Reactive Distillation Process: Production of Lactic Acid by Edreder (2010) develops a model for esterifying lactic acid with methanol and distills the methyl lactate, the lactic acid is recovered by hydrolysis. The purity analyzed was 80-99% molefraction, it took 4 refluxes to theretically reach the highest purity.
Recovery of lactate esters and lactic acid from fermentation broth
Recovery of lactate esters and lactic acid from fermentation broth issued to Cockrem et al details a method for continuous recovery of lactic acid from fermentation broth using reactive distillation with an alcohol.
Solvent Extraction
http://www.google.com/patents/US4771001 Production of lactic acid by continuous fermentation using an inexpensive raw material and simplified form of purification issued to Bailey et al on September 13 1988 deals with a method for production of lactic acid from a the raw sugar source whey permeate and purification solvent extraion. The operational advantage of such a system is the high cell densities that can be achieved and maintained however this raises the challenges of preventing fouling particularly for membrane based cell recycling.
Polylactic acid polymerization
Melt–solid polycondensation of lactic acid and its biodegradability
Melt–solid polycondensation of lactic acid and its biodegradability T. Maharanaa, B. Mohantyb, Y.S. Negi examines the developments in polycondensation formation of PLLA with a set of 4 catalysts. With a decade of research into the technique the reaction dynamics and structure of different stereoisomers has been examined by many investigators. The polycondensation polymerization routes is being investigated for its lower cost and capital investment over the ring opening procedure despite initially producing a polymer of less desirable parameters, such as high melt viscocity, discolorization, and difficulty achieving high molecular weight products. Tin oxide formed from tin chloride with a protonic acid (such as p-toluenesulfonic acid - TSA) is a potent catalyst that can be used in such low concentrations that purification isn't necessary. The polymerization mechanism model originally proposed by Moon involve the coordination of terminal carboxyl condensation with the hydroxyl group on the center carbon. TSA may not be catalytically involved in the reaction, but it may occupy a tin catalytic site to increase the favorability of the desired reaction over side reactions. The reaction can achieve high molecular weight PLLA (ca. 105 Da) by the catalysis of tin(II) chloride dihydrate with an equimolar amount of TSA within 35 h under 0.13–2.66 kPa pressure and within a temperature range of 180–200 C with an average yield of 67%. TSA evaporates during the process and a second addition can further assist the reaction. Solid state polymerization uses temperatures above Tg, but below Tm and sometimes plasticizers to increase terminal end mobility (reaction substrate) to form PLA of high MW with desirable properties.
Syntheis and Properties of High Molecular Weight Poly(Lactic Acid) and its resulting fibers
Syntheis and Properties of High Molecular Weight Poly(Lactic Acid) and its resulting fibers by Zhang and Wang tests the polylactic acid polymerization melt/solid polycondensation process with a number of catalysts. The now commercial route uses a first step of azeotropic dehydration with reflux, in a high boiling point, aprotic solvent like diphenyl ether to produce 50 kDa polymers. The second step joins these fibers with reduced pressure and a tin and protic acid catalyst. This study uses SnCl2·2H2O/p-toulenesulfonic acid monohydrate (TSA) and SnCl2·2H2O/maleic anhydride catalyst for the first step and TSA in the second step. This improves upon Moon et al. with the addition of TSA in the second step because the environmental polarity was found to be shifted in the first step. Polymer MW had leveled off in step 1, but it continued in step 2 with the addition of more catalyst (TSA). SnCl2·2H2O/maleic anhydride was found to be a more effective catalyst due to the less crystalline nature which allowed further polymerization in the amorphous regions. An increase in reaction temperature for the second step was found to be effective up to 180 C and degradative at higher temperatures. The process starts by combining 400 g distilled lactic acid is with the designated catalyst (0.5% wt SnCl2·2H2O and 0.4% wt TSA or maleic or succinic anhydride)and sealed in the reactor. The reaction is heated to 150 C 4 hrs , the reaction is then heated to 160 C and the pressure reduced to 500 Pa for 4 hrs. After an initial low-weight polymerization the reflux condenser is removed and 0.4% wt (of starting lactic acid) TSA is added to the reactor. The temperature is further increased to 180 C and the pressure reduced to 300 Pa for 10 hrs. The polymerization product was dried and processed used standard melt spinning procedures before a final draw between 150-200 C under nitrogen. The final fiber product was characterized with FTIR, DSC, an Ubbelohde viscosimeter, and tensile-testing machine. Final spinning produced a fiber made of high molecular weight polymers and has high tensile strength.
Melt/solid polycondensation of l-lactic acid: an alternative route to poly(l-lactic acid) with high molecular weight
[http://144.206.159.178/FT/862/34857/596552.pdf Melt/solid polycondensation of l-lactic acid: an alternative route to poly(l-lactic acid) with high molecular weight] by Moon et al (2000) describes a method that yields high weight PLA on the order of 500,000 daltons through a condensation reaction using a tin chloride dihydrate/p-toluenesulfonic acid binary system. They report that reaction temperatures below the Tm (melting point) of PLA yields a better product and is referred to as melt/solid polycondensation. oligo(l-lactic acid) (OLLA) is mixed with tin(II) chloride dihydrate (SnCl2) (0.4 wt% relative to OLLA) and p-toluenesulfonic acid (TSA) (an equimolar ratio to SnCl2). The mixture is heated to 180 C and the pressure reduced to 10 torres over the period of an hour followed by maintenance for 5 hrs. The product consisting of 20,000 dalton polymers is ground and heated to 105 C under vacuum for 1-2 hr to crystallize the polymers. Solid-state post-polycondensation was initiated by increasing the temperature to 150 C and reducing the pressure to 0.5 Torr. Treatment was continued over 30 hours, but molecular weight peaked between 2-10 hours and drastically reduced after 20 hours. The results showed a method to obtain high molecular weight PLLA with comparable characteristics to the lactide ROP synthesis. The method used here catalyzes the first step of dehydration to form lactide and the lactide ROP step follows. The high activity of the catalyst and the ability to move through the amorphous PLLA may be driving the reaction by concentrating ester tails and catalyst in the amorphous regions during crystallization.
Basic properties for film polylactic acid produced direct condensation polymerization of lactic acid
Basic properties for film polylactic acid produced direct condensation polymerization of lactic acid by Ajioka characterizes the polylactic acid products of different catalysts and solvents under 130-250 C. Polymerization was commercially pursued from an isolated dilactide intermediate, but direct polymerization is possible due to improvements in kinetic control, removal of resulting water, and suppression of depolymerization. Solvents controlled the rate of reaction based upon their boiling point and the ability to remove water and a Dean Stark trap used, diphenyl ether results shown. Tin and protonic acids catalysts were found to have superior performance with tin(II) chloride achieving highest efficiency and high molecular weights. Zinc catalysts produced maximum 150 kDa weight polymers at 160 C. Weights and flow rates comparable to dilactide process and usable for injection molding. D and L enantiomers were polymerized in ratios of 50/50 to 0/100 respectively. Pure L form had the highest strength and molecular weight. 13C NMR of direct condensation PLA showed 5 carbonyl signals, an additional lower signal from adjacent L subunits.
Direct synthesis of PLA general protocol
In a reaction chamber with a Dean Stark trap 40.2 g 90% lactic acid and 0.14 g tin were dissolved in 400 ml organic solvent for 2 hr at 140 C. The trap was replaced with a tube containing 40 g molecular sieve (3 A) for azeotropic separation for 20 to 40 hr at 130 C. At half volume 300 ml chloroform was added and catalyst removed with filtration or extraction. PLA product was by precipitation by 900 ml methanol and washing over suction with methanol.
Cyclic oligomer production
10.0 kg of 90% lactic acid azeotropically dried 81.1 kg diphenyl-ether organic solvent with 6.2 kg tin catalyst at 150 C for 2 hr. This was followed by recycling of solvent using 4.6 kg molecular sieve (3 A) for 40 hr at 140 C. The reaction was concentrated to 70 kg, cooled to 40 C, and PLA crystals collected. The reaction was concentrated to 5.8 kg. A 11.6 kg hexane was added to the filtrate and an oil separated with 5.8 kg acetonitrile and 1 M HCl. An oily substance was collected after 30 min of agitation and washed with 5.8 kg water. The washed product was dissolved in 2.9 kg chloroform and combined with 4 l isopropyl alcohol and a final precipitated product collected over suction and dried with reduced pressure. Final yield 350 g.
Synthesis of polylactic acid by direct polycondensation under vacuum without catalysts, solvents and initiators
Synthesis of polylactic acid by direct polycondensation under vacuum without catalysts, solvents and initiators by Achmad et al details a procedure. The authors recommend PLA be pursued using processing plants capable of fermenting feedstock, purifying lactic acid, and condensing the product as is proposed for the OSE product ecology. Streptococcus bovis is a LAB suggested for use, but the species is also linked to pathogenicity. The process used by the researchers used three phases for treating the lactic acid and polymerizing its monomer: distillation, oligomerization, and polymerization. The reaction was carried out in 4 l sealable flasks, on magnetic stirrers and heaters, with temperature and pressure probes, and connected to a pressure regulator. During distillation sample temperature is brought to 150 C over 90 minutes and maintained for 60 minutes and PLA concentration increases from 90% to 100% as measured by acid-base titrations. Oligomerization phase was a reduction in pressure to 10 mmHg and temperature was raised to 200 C. The polymerization step was maintenance of the reduced pressure and temperature for 89 hours. The condensate was also separated with gel filtration chromatography and measured with a RI detector. Fourier Transform Infrared Spectroscopy was used to analyze molecules functional groups.
Stereoselective Polymerization for a racemic monomer with a racemic catalyst Direct Production of the polylactic acid stereocomplex from racemic lactide
http://www.cem.msu.edu/~smithmr/Publications/ja9930519.pdf Stereoselective Polymerization for a racemic monomer with a racemic catalyst Direct Production of the polylactic acid stereocomplex from racemic lactide by Radano et al uses a stereoselective catalysts to polymerize L(+)- and rac-lactide to comprehensive products tacticity. Tacticity is the stero-relation between adjacent chiral subunit (same side or different side). Tacticity has important effects on the polymers interactions and crystallinity, and Poly L(+)lactic acid has a Tm = 180 and the poly rac-lactic acid Tm is near room temperature. Tsuji et al first noted the Tm of combined stereoregular L and D polymers was raised almost 50 C. The stereoselective catalyst was a Schiff base aluminum alkoxide.
A Highly Active Zinc Catalyst for the Controlled Polymerization of Lactide
A Highly Active Zinc Catalyst for the Controlled Polymerization of Lactide by Williams et al (2003) details a Zinc alkoxide compound paired with a ligand that has high reactivity and high molecular weight products. Zinc is an attractive catalyst due to low cost, but it has complicating aggregation behavior. Ligands help prevent this behavior and modulate desirable characteristics; the ligand (HL) used for this study was made by refluxing N,N,N′-trimethylenediamine, paraformaldehyde, and 2,4-di-tert-butylphenol. The ligand was reacted with Et2Zn to yield LEtZn which was then reacted with EtOH to produce LZnOEt the zinc alkoxide catalysts. The catalysts structure was examined in solid state (X-ray crystallography, mass spectrometry) and catalytically relevant solution state (NMR, PGSE) and was found to be dimeric in solid state and monomeric in solution state. This information allows rate equations to be solved. The PLA MW was measured with SEC-MLS and monitored with 13C NMR. The catalyst was found to be effective in L/LZnOEt ratios up to and at concentrations as low as 0.7 mM, higher than any other zinc catalysts reported. The active site is the ethoxy group and reaction is sensitive to exchange agents.
Sources still to be summarized
2020 catalysts
https://link.springer.com/article/10.1007/s10965-016-0976-7
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6539807/
https://www.sciencedirect.com/science/article/abs/pii/S0960852410009508?via%3Dihub
Old
http://www.imm.ac.cn/journal/ccl/1208/120803-663-01061-p2.pdf
http://193.146.160.29/gtb/sod/usu/$UBUG/repositorio/10281082_Lonnberg.pdf
http://www.ch.ic.ac.uk/marshall/4I11/Coates2000.pdf
http://www.ch.ic.ac.uk/marshall/4I11/Coates2002.pdf
Polylactic acid value adding
(Poly)lactic acid: plasticization and properties of biodegrable multiphase systems
(Poly)lactic acid: plasticization and properties of biodegrable multiphase systems by Averous (2001) experimented with measuring the properties of PLA prepared with different plasticizers. Plasticizers included: glycerol, polyethylene glycol, citrate ester, PEG monolaurate, and oligomeric lactic acid. various mixtures of PLA with thermoactive starch polymers (TSP) were prepared and tested. Plasticizer treated samples show a decrease in Tg (glass transition temp) and therefore Tm (melting temp). Oligomeric lactic acid followed by low molecular weight polyethylene glycol were effective plasticizers while glycerol was ineffective. Finding effective methods to combine PLA and TSP would enhance the product.
Processing and Mechanical characterization of plasticized Poly lactide acid films for food packaging
[http://www.e-polymers.org/journal/PAT2005ePolymers/page/Oral%20Presentations/Section%20B/Martino_Ver_nica_Patricia.pro.1728860278.pdf Processing and Mechanical characterization of plasticized Poly (lactide acid) films for food packaging] looks at the use of 4 plasticizers to increase beneficial characteristics for film.