Polyethylene from Ethanol/Manufacturing Instructions
| Polyethylene from Ethanol | ||
|---|---|---|
| Home | Research & Development | Bill of Materials | Manufacturing Instructions | User's Manual | User Reviews | File:Polyethylene from Ethanol.png | |
Ethanol production
Ethanol can be purchased or produced on-site
Construction of fluid bed reactor
Instructions for construction of fluid bed reactor can be found on its own wikipage.
Ethanol dehydration
It has been known in the art that aluminium (III) oxide (AlO3) is capable of catalyzing the dehydration of ethanol to ethylene with heat, and the addition of transition metals can assist the reaction. Chen et al conducted a comprehensive study on transition metal doped aluminium catalysts. They found gamma aluminium oxide doped with Ti02 to have 99%+ yields and selectivity and be superior to other metals. An adapted protocol is below.
Catalyst preparation protocol
Protocol for preparation:
1. Combine 4 kg (approximately 1 l) g-Al2O3 with 5.0 l 0.5 mol/L Na2CO3 (264.9 g) solution in a three-necked rounded-bottomed flask.
2. Calculate 10% wt content for TiO2 and solve for titanium sulfate addition(4 kg * .1 = 400 g/79.8 g/mol TiO2 = 5.01 mol = 5.01 mol * Ti(SO4)2 240 g/mol = 1202.37 g Ti(SO4)2). Add 26.4 l 0.19 mol/L Ti(SO4)2 aqueous solution and 26.4 l 0.5 mol/L Na2CO3 solution are added simultaneously with high agitation. Maintain the solution between pH 7-8.
3. The sulfate precipitate is washed at 4000 g over to remove the sulfate solution. The solution is then dried at 110 for 8 hr and then calcined at 500 C for 4 hr.
4. The calcined catalyst is crushed and sieved through 30-50 mesh.
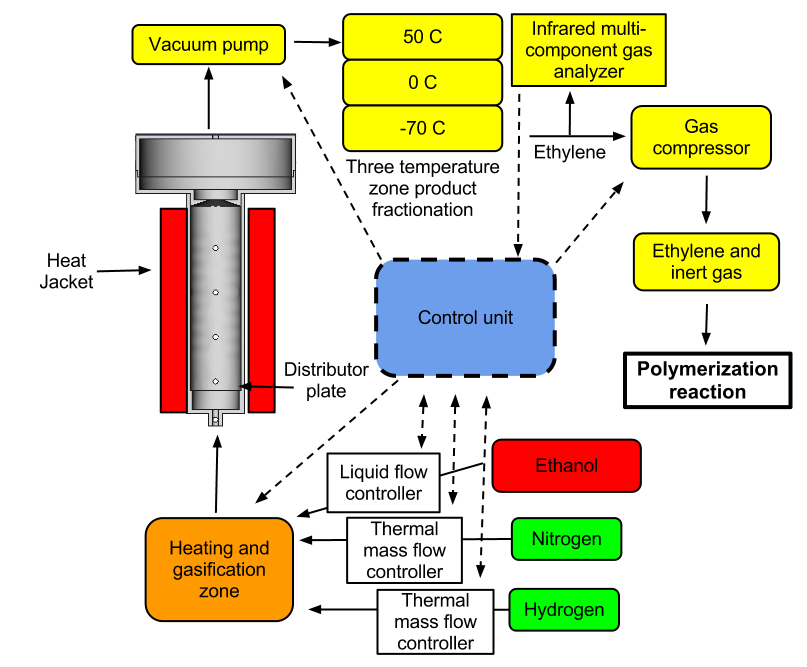
Reactor configuration
Dehydration reaction protocol
1. The reaction zone is filled with catalyst, sealed, and connected to the subsystems.
2. A temperature of 420 - 440 C is established by the heat jacket and a light nitrogen flow is maintained.
3. Ethanol is loaded into the vaporization chamber and heated to 200 C with a Liquid Hourly Space Velocity of 50-100 v/v/hr.
4. Gas products flow through the three temperature phase liquid condenser and liquid water is removed and gaseous ethylene stored.
Ethylene polymerization
An OS Ziegler-Natta catalyst as reported by Goeke is proposed as a first generation polymerization catalyst that is economical and robust. Ti/Mg based catalysts are the foundation of much of the polymerization industry, requiring an electron donating solvent (tetrahydrofuran THF), and an activator of triethylaluminium (TAE). Impregnation of catalyst on a silica base increases the interaction of the substrate and catalyst, and increasing activity of the catalyst. Comonomers may also be included and terminal alkenes are useful for terminating polymer lengthening and controlling product density. Reactor beds may be primed with a polymer to help initiate polymerization and allow removal of product from the reaction bed soon after startup.
Catalyst preparation
1. In a 5 liter flask combine anhydrous 41.8g (0.438 mol) MgCl2, in 2.5 l tetrahydrofuran (THF). Add 27.7 g (0.184 mol) TiCl4 in dropwise over 30 min with stir bar agitation. Add heat ~60 C for 30 min to completely dissolve TiCl4. THF 66 C boiling point
2. Dry 500 g silica at 800 C for 2 hr. Combine with 25 g triethylaluminum in 2 l THF at 60 C for 15 min. Dried activated silica at 65 C.
3. Impregnate support by combining 500 g porous silica with catalyst precursor mixture at 60 C. Dry the mixture at 60 C under N2 for 3-5 hr. Find method to control drying and ED content.
4. Prepare triethylaluminum in isopentane in 5-30% (v/v). Add to heated and loaded reactor.
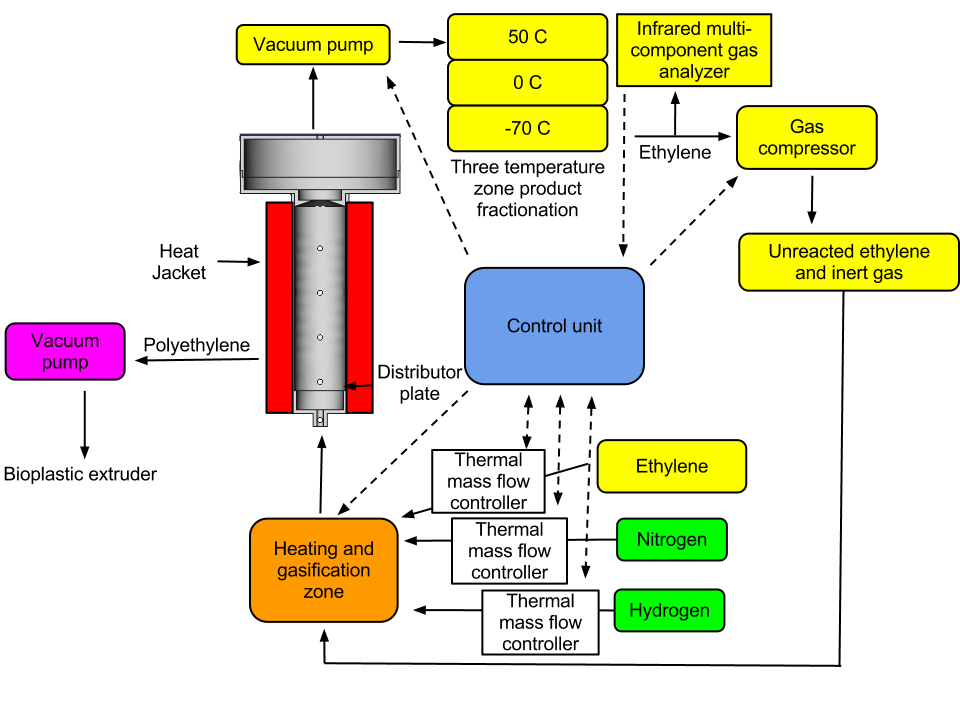
Reactor configuration
Polymerization reaction protocol
1. The reactor is filled with the activated catalyst (~500 ml) on a silica support and an electron donating solvent such as tetrahydrofuran (THF).
2. A temperature of 95-100 C is established and the reactor pressurized to 150-350 psi.
3. Ethylene is added to the reactor in a mixture of nitrogen and hydrogen in a molar ratio of (5:3:2) with a gas flow of 2 m/s. TAE (5%) is added to the reactor in a molar ratio of 15-40 Al/Ti and 2-10% of the reaction mixture. A terminal alkene of C3-C8 is added to the mixture in the amount of 2-10% to control polymer density and acts to terminate chain lengthening.
Controlling the density of the polymer is important to making products suitable for different applications. A low percentage (>2%) of comonomer will not terminate many polymers and result in a high density polymer more suitable for injection molding. A high percentage (2-10%) of comonomer will terminate many polymers and result in a low density polymer suitable to being drawn into a film.
4. Polythylene product is removed at the rate of formation as determined by the loss of ethylene in the recycle stream.